What is Planning of Changes in ISO 9001?
Change Managenent is intended to be beneficial and lead to progress, but should only be implemented when determined by your organization as relevant and achievable.
In addition, you must identify the risks and opportunities or benefits associated with the change.
In order to realize the benefits associated with the identified risks and opportunities, subsequent changes may be needed. These changes could relate to any aspect of any process, such as:
- Inputs
- Resources
- Personnel
- Activities
- Controls
- Measurements
- Outputs
Start with Expert Templates, then Make Them Yours
How Do You Plan A Change Process?
You should seek and record evidence that your organization has retained documented information relating to planning and implementing changes that impact upon the QMS.
To achieve the benefits associated with changes, your organization should consider all types of change that may occur. These changes may be generated, for example, in:
- Processes and procedures
- Documented information
- Infrastructure
- Tooling
- Process equipment
- Employee training
- Supplier evaluation
- Stakeholder management
- Interested party requirements
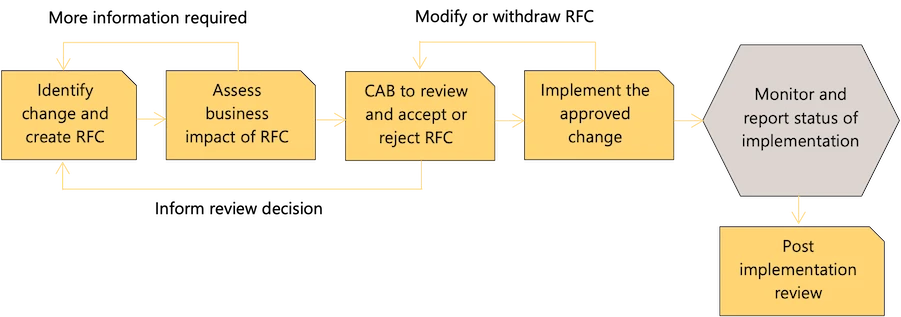
Integrating RFCs (Request For Changes) to the QMS
Ensure that the organization has planned how to integrate and implement the changes into their QMS processes. Check that your organization has considered:
1. The reason for the change; e.g. context of the organization (see 4.1), needs of interested parties (see 4.2), customer feedback, complaints analysis, audit results, performance trends, risk reduction or realizing an opportunity (see 6.1), continual improvement, organization growth, launch of new products/services, organization restructuring etc.
2. Assessing the purpose of the change(s) and potential impact using a risk-based thinking approach to ensure the integrity of the QMS is maintained i.e. focus on priorities, avoid disruption, ensure business continuity, maintain product/service reliability, protect the customer, maintain capability, continue to meet internal/external requirements etc.
3. The resources required to enable the change such as people, knowledge acquisition, infrastructure, environment, budget, trials/tests, ongoing monitoring, structured reviews etc.
4. Determination of responsibility and authority for the change e.g. process owner, lead process users, end users etc., including the necessary communication, training and ongoing review to ensure the change is effective (i.e. the planned activities continue to be realized and planned results are being achieved).
Related Information You Might Find Useful
- 8.1.3 Management Of Change [ISO 45001]