What Are Quality Objectives For ISO 9001?
Quality objectives help to translate organizational purpose (e.g., context and policy) into actionable elements that can be managed and monitored.
The quality objectives and the resulting performance indicators and targets should be factored into your strategic planning to facilitate the integration of the quality management with your organization’s other business processes.
Contents
Don’t Try to Manage It All Alone!
How Do I Develop The Quality Objectives 6.2.1?
Your organization’s quality objectives should be developed to reflect what it does, how well it performs, and what it wants to achieve. When establishing the management system and planning for change, ensure that risks to achieving the quality objectives are identified and the quality objectives are suitably cascaded throughout your organization.
To develop your quality objectives, we recommend businesses establish a cross-functional team (CFT), including key stakeholders, such as the quality manager, top management, and process owners. The Cross Functional Team should be responsible for meeting regularly to discuss the quality objectives and their performance and compliance:
- The initial development and review of quality objectives, targets, and programmes to reflect changes to operations
- Reviewing and monitoring the risk register
- Meeting and reviewing the quality objectives, and communicating any actions
- Reassessing the quality objectives program annually and making any necessary modifications to existing programs and action plans
- Developing new quality objectives programs or determining whether to discontinue existing programs
The outcomes of the Cross-Functional Team meetings should be documented and communicated, and any actions that affect a quality objective should be agreed upon, taken, and followed up promptly.
The Cross-Functional Team's involvement helps build commitment and demonstrates positive leadership. These people are well-positioned to help establish, plan for, and achieve the objectives and meet the targets.
- Get top management to buy in for the quality objectives to ensure that adequate resources are applied and that the objectives are integrated with other organizational goals
- In communicating quality objectives to employees, try to link the objectives to the actual quality improvements being sought, e.g., reducing nonconformances and closing out corrective actions, as this will give people something tangible to work towards
- The quality objectives must be consistent with the overall mission/plan and the key commitments established by your quality policy
- Be flexible with your quality objectives, define the desired result, then let the people responsible determine how to achieve the result
- Objectives can be established to maintain current levels of performance, or to improve performance
- Communicate your progress in achieving the quality objectives and targets across the organization. Consider regular reports on this progress at staff meetings
- How many quality objectives and targets should an organization have? Various QMS implementation projects for small and medium-sized organizations indicate that it is best to start with a limited number of objectives (say, three to five) and then expand the list over time
- Keep your quality objectives simple initially, gain some early successes, and then build on them
- Remember that your suppliers (of services or materials) can help you meet your quality objectives and targets (e.g., on-time delivery)
Ensure that your organization has identified both internal and external issues, and interested parties that are relevant and support the strategic direction of your organization and mitigate aspects associated with issues identified in the:
- Context analysis register;
- Interested party analysis register;
- Risk and opportunity register.
In this way, the strategic direction is utilized to develop the quality policy, the quality objectives, risk management, monitoring and measurement, and the management review processes.

What Types Of Quality Objectives Are There?
There are many types of quality objectives; they can apply to an entire organization, be site-specific, or be specific to individual activities. The appropriate level(s) of management personnel should define the objectives and targets. In some cases, personnel who set objectives may not be the same as those who set targets.
Remember that the objectives are the overall goals as reflected in the principles established in the quality policy.
The differences between strategy, quality policy, objectives, KPIs, and targets are defined below:
- Strategy – Refer to ISO 9001:2015 - 1 & 4.2. What is the purpose of your business? What are you trying to achieve? Typically, a one-line statement may be all that is required. It typically reflects the comments made in the quality policy.
- Quality Policy – Refer to ISO 9001:2015 - 2. This should be a brief statement that shows your top management's commitment to the quality management system. It should also describe the commitment to continuously improving your system's effectiveness. A mention about establishing and reviewing quality objectives relevant to your business needs would also be required. You want to state that your system's success involves all employees' participation.
- Quality Objectives – Refer to ISO 9001:2015 - 2. Set by the Top management in your company, these need to be 'SMART' goals, i.e., Specific, Measurable, Accountable, Realistic, and Time-based. What is it that you are trying to achieve through your processes?
- Indicators/KPIs – Refer to ISO 9001:2015 - 4.1c. These complement your quality objectives. By way of policy deployment, the objectives reflect the quality policy, and the targets reflect the objectives. Indicators or KPIs represent the current status of each goal or target, or the status of operations, management, or other conditions.
- Goals/Targets - generally come from the quality objectives set. That is, ‘what do I have to do to achieve my goals? ’ Set minor goals or targets along the way to ensure you stay on track in reaching each goal and meeting business objectives.
Auditors will expect to review a set of interrelated quality objectives, ensuring they are mutually consistent and aligned with your organization's strategic direction.
How Do I Establish The Quality Objectives?
Organizations must establish the quality objectives at relevant functions and levels, considering the organization’s risks and opportunities. Those objectives must be:
- Consistent with the quality policy;
- Measurable;
- Take into account applicable requirements;
- Relevant to the conformity of products and services and to the enhancement of customer satisfaction;
- Monitored;
- Communicated;
- Updated as appropriate.
We suggest you use your organization’s risk profile to formulate the objectives and goals, which measure the effectiveness of risk mitigation. The related risks must be reviewed before setting the objectives and goals.
The methodology for identifying objectives is as follows:
- Using the risk and opportunity register, identify the top 5 to 10 risks (your risk profile)
- Develop quality objectives to target these top 5 to 10 risks
- Develop goals to meet the objectives
- Develop Key Performance Indicators (KPI) to measure progress towards meeting the goals
- Regularly monitor key performance indicators to ensure progress is being made
A key addition in the 2015 revision of ISO 9001 is using indicators to monitor the achievement of quality objectives.
- The objectives take the goal(s) stated in the quality policy and turn these into statements for improvement against which plans can be made and targets assigned
- Quality objectives may be established to measure the performance of products, processes, customer satisfaction, suppliers, use of resources, and the overall performance and effectiveness of the QMS
- If you state in your quality policy that you will “meet customer requirements”, then you might set customer-focused objectives for: product defects, customer complaints and returns, on-time delivery, etc.
Indicators or KPIs are defined as a measurable representation of the status of operations, management, or conditions. Each objective will need one or more associated KPI and target.
How Do I Create SMART Objectives?
Quality objectives must be specific and measurable to give clear direction as to what is required and the expected outcome, often called SMART (Specific, Measurable, Attainable, Realistic, and Time-based) objectives. Below are suggestions for categories of quality objectives:
- Product: reduction in defect rates, PPM, scrap rates, and on-time delivery
- Process: improving productivity, reduction of waste, set-up times, or rework, improved cycle times
- Customer: product returns, reduction in complaints, improvement in customer satisfaction scores, improved on-time delivery
- Suppliers: reduction of complaints or defects, improved on-time delivery
- Resources: availability, capability, personnel, competency, efficiency, absenteeism
The objectives and targets must be consistent with the quality policy. Depending on your organization's size, management structure, and other factors, various personnel may establish and review the objectives with direct Top Management input.
How Do I Create a Register of Objectives & KPIs?
Use the aspirational statements from your quality policy to create the objectives and register them with the related quality objective(s) and the relevant functions and levels while considering the organization’s risks and opportunities.
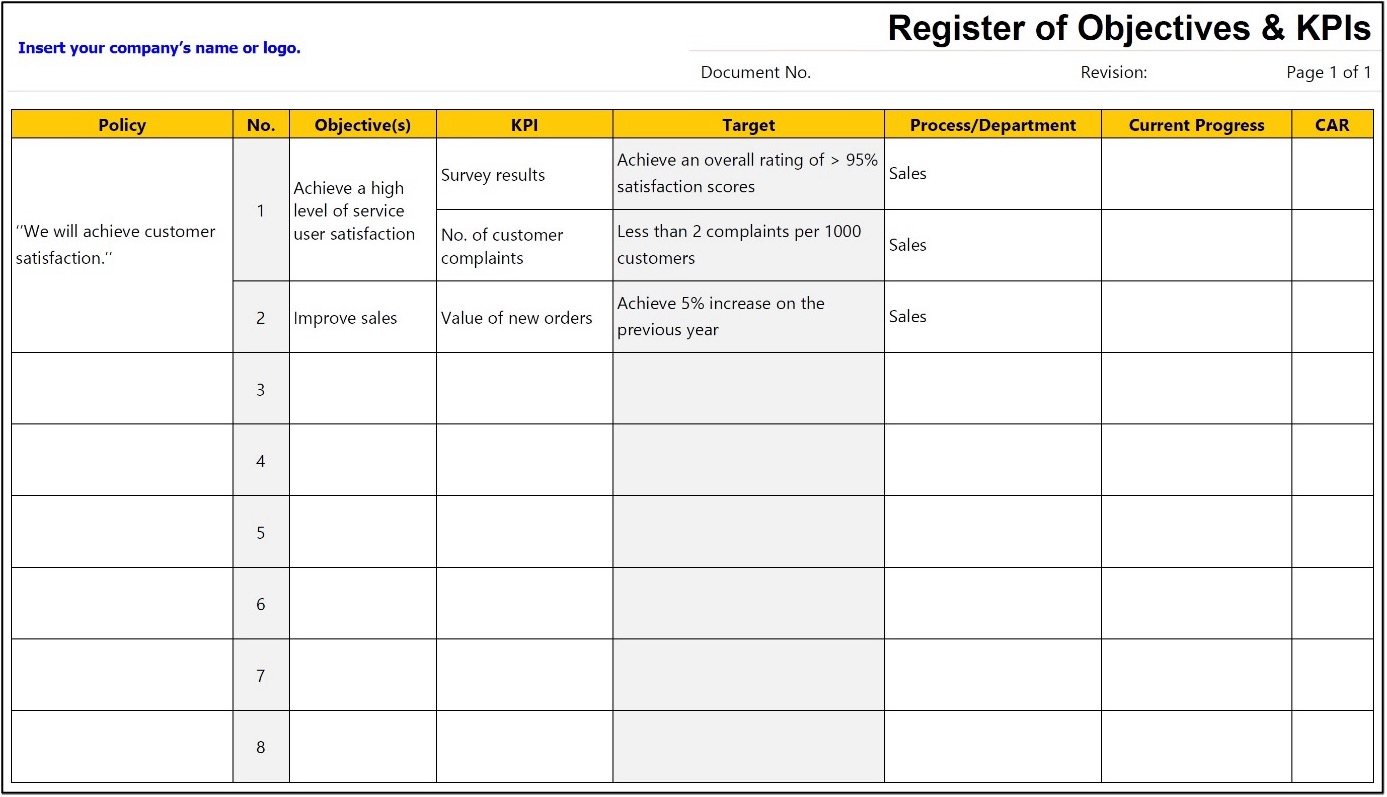
The table below provides ideas for performance indicators that, in turn, may be used to measure the achievement of each quality objective.
| Quality Policy | Quality Objectives | Performance Indicator |
|---|---|---|
| ‘We will achieve customer satisfaction’ | Achieve a high level of service user satisfaction | Survey results |
| No. of customer complaints | ||
| ‘By providing reliable systems’ | Maintain a high level of system reliability | The time system is down during regular work hours |
| ‘That meets customer requirements’ | Ensure systems provided are fit for purpose | Survey results |
| User agreement after completion that the request meets the original specification | ||
| ‘We will respond as quickly as possible when contacted by a customer, and we will work to meet agreed deadlines for all data requests’ | Provide a responsive customer service | Compare actual request completions against the targets agreed with users |
| ‘In the event of a serious incident, we will restore full system operation in the quickest possible time’ | Maintain the ability to recover the systems in the event of a disaster | Time taken to recover from disaster (conduct annual or bi-annual test) |
When Do I Communicate The Quality Objectives?
Certification auditors expect to see a documented plan or roadmap for communicating and meeting the quality objectives. The auditors will also determine whether the objectives meet the policy's intent and are understood by interviewing personnel at various levels.
Inferred awareness through knowledge of the quality objectives is considered insufficient. If the personnel interviewed do not know their measurable objectives and/or do not know the organizational quality objectives they directly affect, the auditor would be further directed to evaluate Top management’s methods of communicating quality objectives.
Quality objectives must be communicated at the relevant levels within the organization to ensure that teams and individuals are aware of their importance and contribution.
The communication of quality information, including quality policy and quality objectives, is often delivered via:
- Text (e.g., newsletter, email)
- Visual media (e.g., posters, short videos)
- Crew or team briefings
- Intranet websites
- Other means, as appropriate, depending on the size and complexity of the organization
When communicating the quality objectives to your employees, link the objectives to the actual quality improvements sought by the business to give staff something tangible to work towards.
Communication with personnel also provides relevant, up-to-date information about quality objectives, management activities, and critical quality information. This information should be retained as part of the management system’s data.
Start with Expert Templates, then Make Them Yours
How Do I Achieve The Quality Objectives 6.2.2?
How Do I Establish An Action Plan For Each Objective?
Organizations must maintain documented information when establishing action plans to achieve their quality objectives.
When planning how to achieve its quality objectives, organizations must first determine:
- What will be done
- What resources will be required
- Who will be responsible
- When will it be completed
- How will the results be evaluated? Include indicators for monitoring progress toward achievement of measurable quality objectives (see clause 9.1.1)
Organizations should consider how actions to achieve their quality objectives can be integrated into their business processes by getting the different parts of the organization to understand how they each contribute to meeting the quality objective.
This ensures that all process owners who impact the quality objective know what their role is, what they need to do, and when they should do it. For example:
- For on-time delivery to customers, the Purchasing team might have an action item to renegotiate delivery schedules
- The Maintenance team might have an action item to implement an improved maintenance schedule to address excessive equipment downtime
- Operations might take action to complete cross-training of employees to prevent work stoppages when key employees are on leave
Establishing an action plan for each quality objective may require considerable effort from the personnel at relevant levels within your organization. To ensure the progress of the action plan and a coordinated effort, a target leader should be selected for each target.
Once the action plan is established, you must implement it. You may find that the following suggestions will help foster a cooperative effort in accomplishing the plan:
- Involve your employees early in establishing and carrying out the action plans
- Communicate the expectations and responsibilities laid out in the action plans to those who need to know
- Build on the plans and programs you have now for quality management system compliance
- Keep it simple
- Focus on continual improvement of management programs over time
The quality management program should be revised regularly to reflect changes in your organization’s objectives and targets. Track all new or modified operations, activities, and/or products in case the management program needs to be amended to reflect these changes.
The methods used to evaluate the achievement of objectives might include: periodic performance comparison against established targets or expectations during management review, functional reviews, process reviews, departmental reviews, personal development reviews (appraisals), and team meetings.
How Do I Develop An Objectives Management Program?
Organizations must establish and maintain one or more management programs to achieve their objectives. Capture how each quality objective will be achieved by assigning a management program to each objective.

The management improvement program is a key element to the success of the quality management system. Properly designed and implemented, management programs will help achieve the objectives and, consequently, improve your organization’s performance in the long term.
The management program must:
- Address each objective and target;
- Designate the personnel responsible for achieving targets at each relevant function/level of the organization;
- Provide an action plan describing how each target will be achieved;
- Establish a time frame or a schedule for achieving each target.
The programs are completed and documented logically, with objectives, targets, and tasks building upon each other to reach all the identified objectives and targets. The objectives, targets, and programs are documented and retained.
How Do I Provide Evidence Of Planning?
You should seek and record evidence that effective planning was undertaken to support the organization’s quality objectives and achievement.
For each quality objective and target, one or more tasks provide a step-by-step description of how the objective and target will be achieved.
- The objectives, targets, and programs are documented
- Each quality management plan designates the responsible position or authority for each task listed
- Timescales for achieving objectives are documented, e.g., stepped achievement (weekly, monthly, quarterly), specific milestones (defined dates or periods), and annual achievement
- Relevant functions and levels of the organization necessary to complete tasks are addressed
- Responsibility for achieving objectives at the various levels within the organization, e.g., top management, functional leadership, department leadership, process owners, teams, and individuals
- Methods used to evaluate the results, e.g., periodic performance comparison against established targets or expectations during management review, functional reviews, process councils, departmental reviews, personal development reviews (appraisals), and team meetings
You should ensure that the planning activity takes into consideration Clause 6.2.1, as well as the following points:
- Identification of processes, resources, and skills needed to achieve quality
- Identification of suitable verification criteria at appropriate stages
- Compatibility of design, production, inspection, and testing
- The confirmation of the criteria of acceptability for all features and requirements
- Details of calibration of any special measuring or test equipment to be used
Do I Need To Document The Quality Objectives?
Your organization must set objectives for relevant functions, levels, and processes within its QMS. Your organization must decide which functions, levels, and processes are relevant.
We recommend you document your objectives using a description or matrix. In addition, it provides a time frame for achieving the objectives and sets out indicators to monitor the achievement of the objectives.
Indicators are defined as a measurable representation of the status of operations, management, or conditions. Each objective will need one or more associated performance indicators.
Maintain documented information to demonstrate that the quality objectives are:
- Consistent with the quality policy (Refer to ISO 9001:2015 - 2.1) to ensure that they underpin the strategic direction of the organization, support the needs and expectations of interested parties, and enhance customer satisfaction
- Aligned to applicable requirements, e.g., those defined by the organization, customer, or regulator, and relevant to the conformity of products and services
- Monitored using suitable means to ensure that the quality objectives are being met, e.g., Business Plan Deployment (BPD) charts, dashboards, matrices, reports, progress charts, traffic light charts, management review, etc.
- Communicated at the relevant levels within your organization to ensure that teams and individuals are aware of their importance and contribution
- Updated accordingly to demonstrate progress and to consider changing circumstances that could result in new, expanded, amended, cancelled quality objectives etc.
Documented information on objectives typically includes a description or matrix of the quality objective and the corresponding means and timeframe to achieve it.
How Do I Audit The Quality Objectives?
Any internal audit of your organization’s QMS should include a review of current internal performance against quality objectives, KPIs, and targets, including continual improvement activities and associated corrective action or management review records.
Verify that your organization’s overall quality objectives:
- Have been defined
- Reflect the quality policy
- Have been translated into goals and indicators
- Are substantially coherent
- Are aligned and compatible with the organization’s context and strategic direction
- Are aligned with its overall business objectives, including customer expectations
If this is not the case, you should evaluate Top management’s commitment to quality. The quality objectives must be measurable and verifiable, but not necessarily quantified. Qualitative results may also be relevant, e.g., a ‘yes or no’ answer regarding achieving quality objectives is acceptable, providing evidence can support it.
There is no specified way of identifying or documenting quality objectives, as these may be present in business plans, management review outputs, annual budgets, etc. Internal auditors should satisfy themselves that the objectives are adequately documented.
You should obtain evidence of how the quality objectives are suitably cascaded throughout your organization’s structure and processes, linking the general strategic objectives to management objectives and specific operational activities.
You should also ensure that the quality objectives are realistic and relevant and that your organization has assigned responsible people the resources needed to meet their objectives. Evidence of this should be obtained throughout all levels of your organization.
Quality objectives are not static and need to be updated in the light of the current business climate and the quest for continual improvement. Internal auditors should verify that your organization's overall performance reflects the aims of the quality policy and reasonably meets the quality objectives.
| Ref. | Quality Objective Requirements | Yes/No | Evidence /Documents |
|---|---|---|---|
| 6.2.1 | Has your organization established quality objectives at relevant functions, levels, and processes needed for the QMS? | ||
| Are the quality objectives consistent with the quality policy? | |||
| Are the quality objectives measurable? | |||
| Do the quality objectives take into account applicable requirements? | |||
| Are the quality objectives relevant to the conformity of products and services and the enhancement of customer satisfaction? | |||
| Are the quality objectives monitored? | |||
| Are the quality objectives communicated? | |||
| Are the quality objectives updated as appropriate? | |||
| Does your organization maintain documented information on the quality objectives? | |||
| 6.2.2 | When planning to achieve its quality objectives, does your organization determine what will be done? | ||
| Does your organization determine the required resources when planning to achieve its quality objectives? | |||
| When planning to achieve its quality objectives, does your organization determine who will be responsible? | |||
| When planning to achieve its quality objectives, does your organization determine when they will be completed? | |||
| When planning to achieve its quality objectives, does your organization determine how the results will be evaluated? |
How Do I Establish The Performance Indicators?
A key addition in the 2015 revision is using performance indicators to monitor your processes and their effective operation, and to achieve the quality objectives. (Refer to ISO 9001:2015 - 4.4.1c). Each quality objective may require one or more associated indicators to provide enough detail about its performance.
Quality management system performance indicators are defined as a measurable representation of the status of operations, management, or conditions. Each objective may require one or more associated quality performance indicators and target.
Performance indicators are generally data-based expressions of the frequency of occurrence of events, incidents, or reports. These performance targets are measured and monitored in conjunction with performance indicators, expressed in ratios, averages, moving averages, rates, or trends as appropriate.
The standard requires your organization to determine and apply the criteria and methods to ensure effective operation and control of the identified processes. Such criteria and methods might include, for example, process monitoring indicators, process performance indicators, target setting, data collection, performance trends, or internal/external audit results. Other criteria and methods might include:
- Arrangements for governing each process, e.g., process owner reviews, process committees, flight deck/dashboard representations, risks and opportunities relating to the process, resource needs, user training/competency, continual improvement initiatives, frequency of reviews, agenda, minutes, actions
- The organizational approach towards continual improvement and the types of action taken when process performance is not meeting intended results
- Identifying ‘what' needs to be monitored and measured to determine QMS and process performance, e.g., Internal - achievement of quality objectives, process performance indicators, target setting, audit results (conformity/nonconformity, effectiveness), trends. External - customer and supplier performance, e.g., survey results, satisfaction indicators/ratings, compliments, complaint profile, returns/rejections, warranty claims, invoice queries
Objective evidence should indicate that your organization has successfully integrated the QMS processes into its business processes. Evidence may include top management reviewing QMS KPIs as part of regular business reviews, awareness of contractors and employees of QMS goals and expectations, etc.
Depending on your organization's size, management structure, and other factors, the quality objectives may be established and reviewed by various personnel and with direct Top management input. The objectives, performance indicators, and targets must be consistent with quality policy and business strategy.
How Do I Link The Performance Indicators To The Objectives?
The performance indicators must link to and support the relevant quality management system objectives. Performance indicators need to be simple, measurable, and reliable. The KPIs include a mix of outcome indicators (e.g., nonconformity rates, % audit success, open/closed status of risk assessments) and process indicators (e.g., validation of quality critical processes, and record keeping).
Performance assessment results are used in two ways:
- To measure the effectiveness of risk mitigation measures by comparing KPIs to targets set in the quality objectives statement
- To identify potentially new risks resulting from ineffective mitigations
A hierarchy of quality targets is formed, from strategic objectives (e.g., to minimise adverse effects), through to operational and more detailed functional objectives (e.g., to increase levels of quality training by a stated amount), to specific performance criteria (e.g., reducing occupational exposure limits or lost-time incidents).
You could create an Excel workbook with tabs for defining the quality objective, an action plan, and the actual data. Stay on top of it with regular updates and reviews to ensure actions are completed to drive improvement. You should consider and implement the following to help ensure compliance with ISO 9001:2015 - 6.2:
- Ensure that quality objectives are established within the relevant functions (Engineering, Purchasing, Finance, Human Resources, Quality, IT, etc.), and are linked to the respective functional strategy, e.g., functional direction, process improvement, milestones etc.
- Ensure that quality objectives are established within relevant processes, which are defined by the process owner relating to the aims of the process, e.g., what the process is trying to achieve, resources needed, timescales etc.
- Ensure that quality objectives are established and suitably cascaded across your organization's structure, e.g., Top management, middle management, supervisors, departments, projects, groups, and individuals
The table below shows the quality objectives and provides ideas for performance indicators and ideas for targets that, in turn, may be used to measure the achievement of each quality objective.
| Policy Statement | Quality Objectives | Performance Indicator | Possible Targets |
|---|---|---|---|
| ‘We will achieve customer satisfaction’ | Achieve a high level of service user satisfaction | Survey results | Achieve an overall rating of > 5.5 this year |
| No. of customer complaints | Fewer than two complaints were submitted during the period. | ||
| ‘By providing reliable systems’ | Maintain a high level of system reliability | The time system is down during regular work hours | Systems downtime < 5% during working hours |
| ‘That meets customer requirements’ | Ensure systems provided are fit for purpose | Survey results | Achieve “fit for purpose” rating of > 5 on survey |
| User agreement after completion that the request meets the original specification | 100% of completed requests signed off to show customer agreement | ||
| ‘We will respond as quickly as possible when contacted by a customer, and we will work to meet agreed deadlines for all data requests’ | Provide a responsive customer service | Compare actual request completions against the targets agreed with users | 95% of work requests were completed within the deadline |
| ‘In the event of a serious incident, we will restore full system operation in the quickest possible time’ | Maintain the ability to recover the systems in the event of a disaster | Time taken to recover from disaster (conduct annual or bi-annual test) | Recover system operation within 6 hours of a disaster |
| ‘Reduce usage of hazardous chemicals’ | Increase the use of non-hazardous chemicals | Amount of hazardous chemicals used versus usage of non-hazardous chemicals | Reduce use of high-VOC paints by 25% by January 2026 |
| ‘Reduce energy usage’ | Reduce electricity use | Electricity billing BMS monitoring output | Reduce usage by 10% annually |
| ‘Zero harm to workers’ | No serious incidents | Incident rates and trends Efficiency of emergency response procedures | Incident rate 2.5% below industry average |
The table also provides ideas for KPIs and targets that, in turn, may be used to measure the achievement of each objective.
How Do I Audit The Performance Indicators?
Use of the process approach by your organization to identify sources of input, activities, output, receiver of output, performance indicators to control and monitor processes, the risks and opportunities associated with them, and an action plan to address them.
The use of indicators needs to be audited in detail:
- Are quality objectives and subsequent indicators based on sound information?
- Are indicators really related to the corresponding quality objectives?
- Are statistical tools needed to define and monitor quality objectives?
- If the indicators reach expected values, how does your organization ensure the quality objective has been achieved?
Your organization should audit the performance indicators in detail to establish whether the quality objectives are based on sound information and whether the indicators and targets are related to the corresponding objectives. Ensure your organization uses statistical tools to define and monitor the quality objectives.
Certification Auditors will expect to review a set of interrelated objectives, ensuring that they are mutually consistent and aligned with your organization's strategic direction.
Often, Certification Auditors will interview members of Top management to determine whether your organization has established and ensured the continued relevance of its quality policy, related quality objectives, indicators, and targets, as well as how performance measures are established, whether they are aligned to your organization’s context, and the requirements of your interested parties.
| Ref. | Management Review Requirements | Yes/No | Evidence /Documents |
|---|---|---|---|
| 4.4.1d | Has your organization determined and applied the criteria and methods (including monitoring, measurements, and related performance indicators) to ensure these processes' effective operation and control? | ||
| 9.1.1 | Does your organization determine what needs to be monitored and measured? | ||
| Does your organization determine the monitoring, measurement, analysis, and evaluation methods needed to ensure valid results? | |||
| Does your organization determine when the monitoring and measuring shall be performed? | |||
| Does your organization determine when the results from monitoring and measurement shall be analysed and evaluated? | |||
| Does your organization evaluate the performance and the effectiveness of the QMS? | |||
| Does your organization retain appropriate documented information as evidence of the results? | |||
| 9.1.2 | Does your organization monitor customers’ perceptions of how their needs and expectations have been fulfilled? | ||
| 9.1.3 | Does your organization analyse and evaluate appropriate data and information arising from monitoring and measurement? |
The objective of Key Performance Indicators (KPIs) is to provide a transparent and comprehensive framework for process control through regular quality assessments of the nonconformance process to evaluate whether the process is running according to expectations in terms of quality, time, and costs, and if changes to the process are required.
What Types Of Performance Indicators Are There?
An organization’s quality management system performance is verified by reference to leading and lagging performance indicators and performance targets linked to our activities, as set by our quality management system in support of our quality objectives.
Leading indicators, which are particularly relevant from a management perspective, may influence management priorities and determine actions for improvement. Leading indicators (proactive) are measured without a nonconformance occurring and help predict or prevent future events linked to processes and activities. Leading indicators that are used include the following:
- Frequency and status of quality audits
- Culture and behaviour
- Attitude and opinion surveys
- Training provided against programs
- Investigations closed within 14 days
- Frequency and status of workplace inspections
Lagging indicators are measures of occurrences, particularly the adverse outcomes that the organization aims to prevent. Lagging indicators are mainly used for aggregate, long-term trending, either at a high level or for specific occurrence types or locations.
Lagging indicators (reactive) are those that show the number and or severity of events that have occurred:
- Lost time incidents
- Nonconformances
- Days lost
- Procedural deviations
- Lower-level system failures and safety events
- Absence data
- Property damage
Performance indicators are intended to be easy to measure and monitor, and relevant to the organisation or workgroup whose performance is being measured, to provide an immediate and reliable indication of the level of performance.
Organizations should ensure that quality performance indicators:
- They are under the control of individuals performing the process
- They are positive and easily understood
- They are relatively easy to monitor
- Consists of a large volume of data that can be fed back
- Reflect on current situations, not historic ones
Performance indicators are understood and owned by the organization or workgroup whose performance is being measured, whilst being cost-efficient in equipment, personnel, and additional technology required to gather performance information.
What Are Performance Targets?
Targets define long-term quality management system performance objectives. They are expressed numerically and must be concrete, measurable, acceptable, reliable, and relevant.
Targets also contain completion dates with milestones if the target is to be achieved in phases or over an extended period. Targets provide a measurable way of ensuring and demonstrating the effectiveness of quality performance objectives.
Targets are based on performance criteria that describe the standards or limits to which a particular activity or system element is to perform. These apply at various levels within our quality management system. Such criteria may establish the nature and frequency of such tasks as:
- Plant maintenance
- Quality management system reviews and audits
- Assessment of training needs
- Hazard and effects identification and risk assessment
- Testing of emergency plans
- Testing emergency shut-down and prevention systems
- Testing fire detection, protection, and alarm systems
- Process and emission monitoring
As the basis for control, monitoring, and performance measurement, criteria are readily measurable and clearly and unambiguously documented.
Can I See Examples Of Key Performance Indicators?
The examples of key performance indicators listed below reflect specific and quantifiable measures that your organization can use to evaluate the quality of the process, ensure ongoing optimization, and process design improvements.
KPIs can be reported quarterly or monthly; however, Top management may require more frequent reporting of metrics and KPIs as necessary. The following performance indicators should be measured over a specific time period, e.g., quarterly, etc.:
| Key Performance Indicator | Definition |
|---|---|
| Purchased parts failure rate | Number of nonconforming parts relative to the total number of purchased parts (per period). |
| Nonconformities raised by the Customer | Number of incoming complaints per x rolling month divided by the number of incoming complaints of the previous reporting period |
| Costs of internal nonconformities | The sum of all costs of rework in relation to the value of the WIP (Work in Progress) |
| Total number of corrective actions | Number of open/closed/overdue corrective actions/total number for a given period/process/lot/batch |
| External providers’ nonconformities | The ratio between the number of supplier nonconformity reports issued to suppliers by the entity during a period in relation to the number of production hours over the same period x multiplied by 1000. |
| Supplier-related nonconformance costs | Nonconformance costs caused by suppliers in relation to total procurement costs. |
| Number of nonconformity reports issued | Ratio between the number of supplier nonconformity reports issued to suppliers by the entity during a period in relation to the number of production hours over the same period x multiplied by 1000. |
| Supplier on-time delivery | The number of parts received from the Supplier divided by the number of parts confirmed to be delivered by the Supplier (per period). |
| Supplier delivery performance | The sum of on-time in the complete purchase order schedule lines matches the sum of expected scheduled deliveries. Note: On-time in full deliveries: Lines were delivered on the planned date +/- x working days. |
The following table provides examples of metrics related to nonconformances. There is no obligation to apply all examples; each organization can define its KPIs and metrics.
| Metrics | Definition |
|---|---|
| Defectiveness after release | Hours spent closing the defects identified after the release in relation to the total effort to develop the service |
| Right First Time (RFT) | Number of deployments meeting the requirements at final inspection/test divided by the number of deployments inspected (per period) |
| Nonconformity processing time | Average time to close all related actions after the opening date (measured over x rolling months) |
| First pass yield | Ratio of "passed" tests over the total number of tests in the last period |
| Complaint processing time | Average time to close all related actions after the opening date (measured over x rolling months) |
| The rate of the total cost of non-quality | Sum of all costs related to scrap, rework, repair, modifications, penalties, and extra warranty costs in relation to sales revenue |
| Claim and warranty costs | Total costs of claim and warranty in relation to sales revenue |
| Nonconformance costs before delivery | Nonconformance costs (e.g., all kinds of unplanned costs) before delivery in relation to the revenue of the service |
How are Quality Objectives Monitored & Revised?
The performance of our organization is measured, including performance indicators and targets linked to our quality management system objectives. The performance of our organization is measured, and the KPIs are continuously monitored and analyzed for trends.
KPIs demonstrate the performance of our organization and the effectiveness of risk controls based on reliable data. KPIs are reviewed and regularly updated to ensure they remain relevant when:
- An indicator shows that risk control is not adequate
- Defining KPIs and targets appropriate to your organization’s activities, risks, aspects, and objectives
- Reviewing whether action has been taken when a KPI shows a negative trend (which may reflect an ineffective risk control or an inappropriate KPI)
- The results of quality performance monitoring are discussed at Top management’s level
- Evidence of feedback provided to the Quality Manager is analyzed and reported
Quality objectives should be monitored using suitable means to ensure that the objectives are being met, e.g., the Business Plan, deployment charts, dashboards, matrices, reports, progress charts, traffic light charts, and management reviews. The objectives must be updated accordingly to demonstrate progress and account for changing circumstances.
Jump Start Your Documentation
Related Information You Might Find Useful
- 6.2 Environmental Objectives and Planning to Achieve them [ISO 14001]
- 6.2.2 Planning Actions to Achieve OH&S Objectives [ISO 45001]